The Shape of Copper
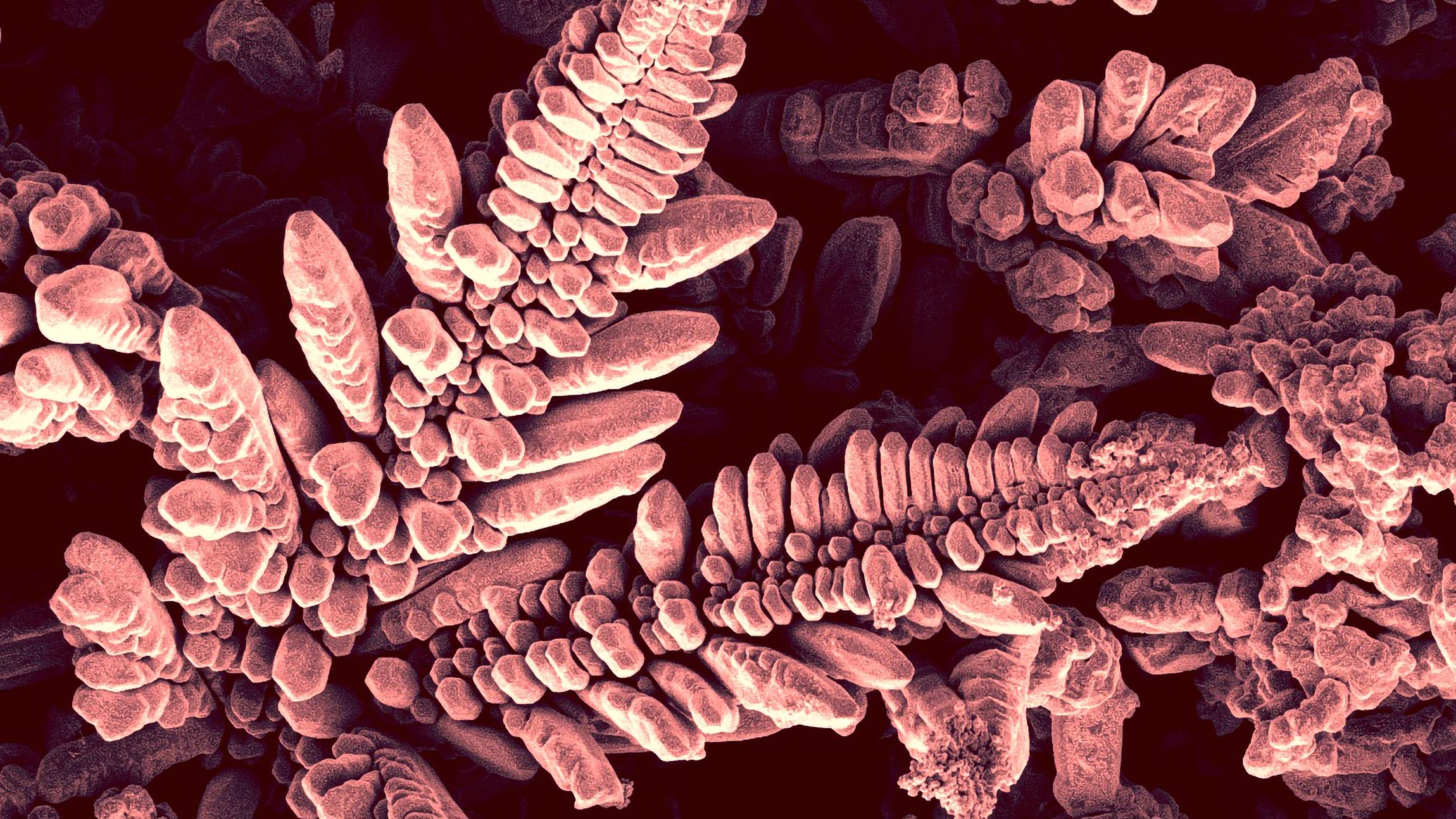
Chemistry Ph.D. student Lamya Tabassum's prize-winning photo of copper sulfide nanoarrays, taken with a Verios 460L SEM electron microscope that can magnify up to 2,000 times, was a happy accident. "I was growing copper sulfide nanoarray on the copper substrate to use it as a catalyst for water splitting," she says. The process produces hydrogen, a valuable energy commodity. Tabassum was scanning the particles under the microscope to see if they actually were arrays.
The strange shape of the nanoarray in this image was not what she was expecting to find. In fact, it was new to science, and now Tabassum is publishing a paper about it.

Lamya Tabassum '22 Ph.D.
We loved the photo Tabassum took, and had so many questions about how and why she took the photo and how it relates to her research at UConn. Here are her answers:
Why are you growing copper sulfide nanoarrays on copper substrate? What is the research for?
I am doing research on renewable energy. My research work involves synthesizing electrocatalysts for water splitting. In electrochemical water splitting, electrodes are placed in water, and electricity is passed through the electrodes to split water into hydrogen and oxygen. The hydrogen produced from water splitting can be stored and used as fuel in industries, vehicles, or fuel cells to produce electricity. Unfortunately, the best electrode materials for water splitting discovered so far are based on precious metals (platinum, ruthenium, iridium). This increases the cost of the watersplitting system. Copper sulfide on the other hand has a much lower cost and has the potential to be an electrode material for water splitting due to its high electric conductivity. Moreover, the nanoarray-like structure can enhance the activity of an electrocatalyst. I was growing copper sulfide nanoarray on the copper substrate to use it as a catalyst for water splitting.
Can you explain what the morphology of the nanomaterials means for a non-scientist audience?
Morphology means the shape, size, structure, and orientation of a material. I work on growing material having nanorod array-like morphology. Basically, in this type of material, an array of nano-sized rod-like features stands on the metal sheet (or substrate).
What sort of research is done at the Thermo Fisher Scientific Center for Advanced Microscopy and Materials Analysis (CAMMA)?
Thermo Fisher Scientific Center for Advanced Microscopy and Materials Analysis (CAMMA) is an outstanding facility for doing research on material science. They have the most advanced electron microscopes here. Optical microscopes can magnify the image only upto 2000 times. Electron microscopes on the other hand can magnify up to 2 million times. With the electron microscope here, it is possible to see features as small as a single atom.
And where is that facility at UConn?
This facility is on the ground floor of UConn's Innovation Partnership Building (IPB).
Can you just tell us a bit more about how special it was to see this for the first time — and why you hadn't been able to before?
My intention was to grow nano-sized rod-like features. But this material showed a different shape. It looked like a fern-like feature was growing on the substrate. To the best of my knowledge, this kind of shape was not observed before for copper sulfide-based nanomaterials. That's why it was so special to me.
Did you pick that color for a reason — do the nanoarrays look like that to the naked eye or when there are a lot of them? I ask because of course they're called copper arrays and the color is copperish — is there any connection?
We can see the nanoarray only under an electron microscope that only gives a black and white image. To the naked eye, it should look dark blue, which is the original color of copper sulfide (covellite mineral). I picked the copper color due to artistic purpose and also because it is a copper-based material.
By Kim Krieger
Photo by Lamya Tabassum '22 Ph.D.
